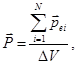|
The electric field in dielectrics
§ 1 The conductors and insulators. Polar and non-polar molecules. Ionic
crystals. Free and bound charges. Types of polarization.
- I. Conductors and insulators, see Lecture 1 on electrostatics.
II. Types of insulators.
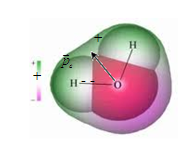
Dielectric molecule as a molecule of any other substance are
electrically neutral. This means that the net negative charge of the
electrons is the sum of the positive charge of the nuclei.
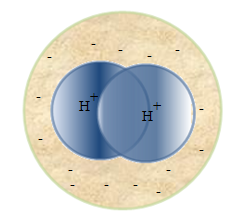
If
the molecules in the absence of an external electric field the centers
of gravity of the positive and negative charges are the same, that
is, the dipole moment of the molecule 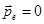 , these molecules are called non-polar. These include molecules H2, O2, N2. , these molecules are called non-polar. These include molecules H2, O2, N2.
Molecules that,
in the absence of an external field the centers of gravity of
positive and negative charges do not coincide, ie, there is a dipole
moment 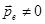 , are called polar. These include H2O, CO, NH, HCl, SO4 etc. , are called polar. These include H2O, CO, NH, HCl, SO4 etc.
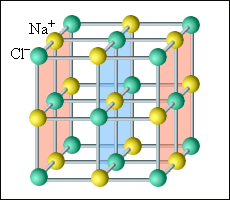
Ionic crystals
(NaCl, KBr, KCl) have a crystalline structure. The nodes of the space
lattice are interleaved ions of opposite signs. In ionic crystals can
not distinguish individual molecules. They need to be seen as a system
of two sublattices - positive and negative.
The crystal lattice of salt
- III. Types of polarization.
Dielectric polarization is the process of dipole orientation or appearance under the influence of an electric field oriented along the field dipoles.
(The appearance of the dipole moment in the dielectric is called polarization)
As a result of the polarization of the molecule acquires a dipole moment 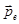 ,which is proportional to the field ,which is proportional to the field
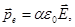
where α - polarizability of the molecule (characterizes the "response" of the molecule to the electric field). α - characteristic of an atom or ion.
As the quantity characterizing the degree of polarization of the dielectric polarization of the vector 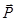 - is taken - the dipole moment per unit volume (or the density of the dipole moment) - is taken - the dipole moment per unit volume (or the density of the dipole moment)
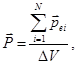
where 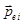 - the dipole moment of a single molecule , - the dipole moment of a single molecule , 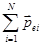 - the total dipole moment of the volume V. - the total dipole moment of the volume V.
Three types of dielectric correspond to three types of dielectric polarization
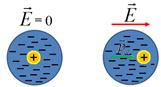 Electronic polarization
- the emergence of the dipole moment in the non-polar molecules.
Electronic polarization due to the displacement of the electron orbit
of the atom relative to the nucleus in an external field . Electronic polarization
- the emergence of the dipole moment in the non-polar molecules.
Electronic polarization due to the displacement of the electron orbit
of the atom relative to the nucleus in an external field .
Ionic polarization
- the emergence of the dipole moment in ionic crystals induced by the
displacement of the sublattices of positive ions along the field, and
negative - against the field . 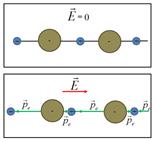
- Orientational (dipole)
polarization - the emergence of the dipole moment in the dielectric
with polar molecules due to the orientation of the dipole moments of
the molecules in the direction of the field .
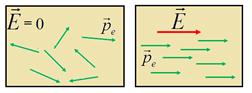
-
IV. Free and connected charges
Charges that when an external electric field can move freely
through a conductor, and are not associated with the ions of the
crystal lattice are called free.
The charges
are part of the molecule, which under the action of the external field
is only slightly displaced from their equilibrium positions, and leave
the molecules can not, said to be linked.
§ 2 Field intensity in the dielectric.
In isotropic dielectric polarization vector is linearly dependent on the field 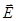
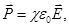
where χ - dielectric susceptibility of the substance, shows how the dielectric response (perceived) to an external electric field.
α - characteristic of a single molecule (or ion), χ - characteristic of the dielectric, ie characteristic matter altogether. χ is independent of 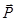 and and 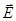
in weak fields. χ – dimensionless
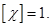
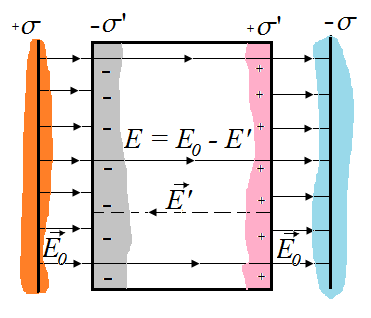 If
between the plates of plate capacitor dielectric layer placed, the
resulting polarization of the positive charges in the dielectric will be
displaced in the field, and negative - against the field, and on the
right side (in the picture) and come into existence the excess of positive there, and on the left side - an excess of negative charges with a surface density + σ 'and -σ'. These charges create inside the dielectric plate a homogeneous field, the intensity of which is equal to the Gauss theorem If
between the plates of plate capacitor dielectric layer placed, the
resulting polarization of the positive charges in the dielectric will be
displaced in the field, and negative - against the field, and on the
right side (in the picture) and come into existence the excess of positive there, and on the left side - an excess of negative charges with a surface density + σ 'and -σ'. These charges create inside the dielectric plate a homogeneous field, the intensity of which is equal to the Gauss theorem
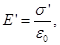
where 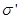 - the surface density of bound charges . - the surface density of bound charges .
Outside the dielectric 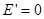 . External field . External field 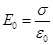 and internal and internal 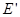 directed towards each other, therefore, inside the dielectric directed towards each other, therefore, inside the dielectric
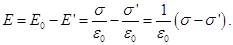
Outside the dielectric 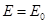 . .
We define surface density of-related charges 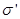 . Total dipole moment of the dielectric plate . Total dipole moment of the dielectric plate
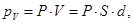
where S - area of ??the face plate, d - its thickness. On the other hand, the total dipole moment is
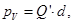
where Q'- bound charge of each face, d - arm dipole .
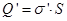 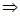
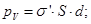
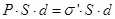
or 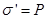
The surface density of bound charges 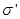 is equal to polarized (polarization) P. is equal to polarized (polarization) P.
Then the field inside the dielectric
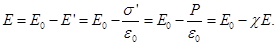
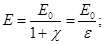
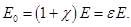
Dimensionless quantity 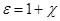 called the dielectric constant. ε
shows how many times the dielectric field is weakened, describing the
quantitative properties of the dielectric to polarize in an electric
field . called the dielectric constant. ε
shows how many times the dielectric field is weakened, describing the
quantitative properties of the dielectric to polarize in an electric
field .
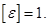
§ 3 The electric displacement.
Gauss theorem for the electrostatic field in the dielectric.
For the description of the electric field, in particular, in the
dielectric, introducing the electric displacement vector (the vector
of electrostatic induction) 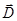 , equal , equal
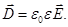
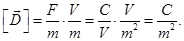
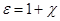 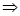 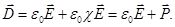
Resulting field is described by a vector in the dielectric intensity 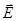 . . 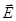 depends on the properties of the dielectric (of ε). Vector depends on the properties of the dielectric (of ε). Vector 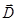 describes
the electrostatic field generated by the free charges. Bound charges
arising in the dielectric, can cause redistribution of free charges up
the field. Therefore, the vector describes
the electrostatic field generated by the free charges. Bound charges
arising in the dielectric, can cause redistribution of free charges up
the field. Therefore, the vector 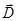 describes
the electrostatic field generated by the free charges (ie in a
vacuum), but with this their distribution in space, which in the
presence of a dielectric. describes
the electrostatic field generated by the free charges (ie in a
vacuum), but with this their distribution in space, which in the
presence of a dielectric.
The lines of force vectors 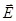 can begin and end as in free, and the bound charges. The lines of force vectors can begin and end as in free, and the bound charges. The lines of force vectors 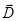 - only free. Through the area of the field where the bound charges, the field lines are vector - only free. Through the area of the field where the bound charges, the field lines are vector 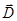 without interruption. without interruption.
Vector flux 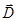 through any closed surface through any closed surface
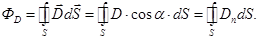
Gauss theorem for the electrostatic field in the dielectric:
The flow of
the displacement vector of the electrostatic field in the dielectric
through any closed surface is equal to the algebraic sum inmates inside
this surface free electric charges:
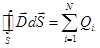
|