main
To the list of lectures
|
Quantum optics. Thermal radiation. §1 the Thermal radiation and the luminescence
2) cathodoluminescence - glow solid when bombarded by electrons (CRT - cathode ray tube in oscilloscopes, CRT TV, etc.); 3) electroluminescence - glow in solids under the influence of an electric field (neon lamps, fluorescent lamps, mercury lamps, arc discharge, LED, etc.); 4) photoluminescence - glow in the absorption of the incident electromagnetic radiation on the body (road signs); 5) scintillation - glow in the absorption of ionizing radiation (scintillation detectors). Luminescence,
which immediately stops at the end of the exciter of light, called
fluorescence, and last for a long time after the termination of the
exciter of light - phosphorescence.
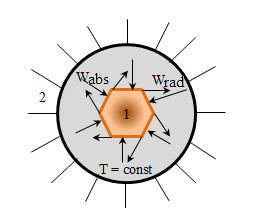
2 - thermostat. Wabs = Wrad - equilibrium thermal radiation.
Thermal radiation is in equilibrium, that is, the energy that is supplied to the body and the body is emitted, are equal. If the emitting body is not getting the energy (heat) from the off, it cools. Thermal radiation is subject to self-regulation. Assume that the body radiates more energy than it absorbs. As a result of its internal energy decreases, therefore, the body temperature decreases, respectively, the radiation intensity decreases, and it will happen as long as the equilibrium process will begin, in which Wrad = Wabs. Processes associated with the establishment of equilibrium thermal radiation explained by the dependence of the intensity of thermal radiation from the body temperature. At low temperatures the bodies emit invisible infrared radiation. With high - red glow. Incandescent body give white glow. Of all the types of radiation equilibrium can only be thermal radiation. By equilibrium processes apply the laws of thermodynamics, so thermal radiation can be described using the laws of equilibrium thermodynamics.
§2 the emissivity coefficient of absorption capacity. Black body
Quantitative characteristic of the thermal radiation is a power spectral density of luminosity - emissivity ability (rν,T) - determines the amount of energy radiated per unit surface area of the emitting body per unit time per unit frequency range from ν to ν + dν
[rν,T] = J/m2 Where rν,T - is a function of frequency and temperature. Used as recording rλ,T - a function of wavelength and temperature. We find the connection between them.
Because λ = с/ν, therefore
Where с - the speed of light, which is equal 3·108 m/s,
The second characteristic of the thermal radiation is absorption - аν,Т, which is also a function of frequency and temperature. Absorption capacity аν,Т (or absorption coefficient) indicates how much energy per unit of time of the incident by one flat surface of the body is absorbed.
аν,Т ≤ 1, [аν,Т] = l (dimensionless). The body, the absorption coefficient is equal to 1 is called absolutely black body (a.b.b.). A black body is able to absorb fully at any temperature all incident radiation on the body at any frequency. Black body not exist in nature, but the black, black velvet, pupil of the eye in a certain frequency range in properties close to the absolutely black body.
Body with a cavity - an example of a black body (аν,Т =1). If аν,Т < 1, and with аν,Т = const, then the body is gray.
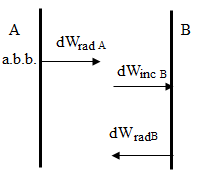
Consequence of Kirchhoff's law: 1) Since for any body аν,Т < 1, from Kirchhoff's law, it follows that the ability of the emissivity of a body rν,Т < rν,Т a.b.b. rν,Т = aν,Т ·rν,Т a.b.b. 2) If the body does not absorb electromagnetic radiation of a certain frequency ν, that is, aν,Т = 0, it is him and not radiate as rν,Т = aν,Т ·rν,Тa.b.b.= 0. Kirchhoff's law describes only the thermal radiation. Radiation, which is not subject to Kirchhoff's law, not the heat - the criterion of thermal radiation. Kirchhoff's law can be obtained by considering the equilibrium thermal radiation. Let two plates, isolated from the environment. In this case, A is the plate of a black body A and B are in thermodynamic equilibrium.
dWinc = dWrad , since there is a thermodynamic equilibrium dWinc В = dWrad А = rν,Т a.b.b. dν ; dWabs B = aν,Т dWinc B = aν,Т rν,Т a.b.b. dν= dWrad B = rν,Т dν rν,Т = aν,Т ·rν,Т a.b.b., therefore, rν,Т /aν,Т = rν,Т a.b.b. Because the surface is selected in a completely arbitrary, the result will be true in the case of any surface. |